Eye surgery
Glaucoma (AMD)
Age-related macular degeneration (AMD)
Cataracts
Operations on the vitreous body
Operations on the conjunctiva
Operations in the event of injuries
Eyelid surgery
Lacrimal duct surgery
Tumour surgery
Glaucoma (Grüner Star)
PD Dr. med. Karl-Georg Schmidt
Definition
Glaucoma, a disease of the optic nerve, is one of
the most frequent causes of blindness in industrialised nations.
Glaucoma is usually painless, insidious, and at first imperceptible.
Characteristic is a continuous irretrievable loss of optic nerve fibres.
These nerve fibres are responsible for forwarding of visual stimuli to
the brain. The more nerve fibres are lost, the less visual stimuli the
brain receives from the eye for image processing. As a result,
characteristic blind spots occur, which untreated will lead to
blindness.
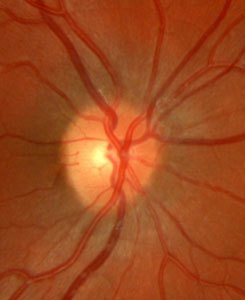
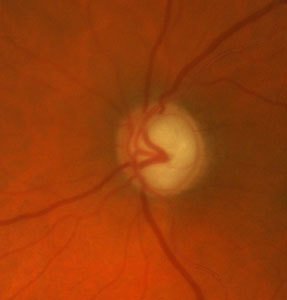
This loss of optic nerve fibres is detectable in the optic nerve head (papilla) at an advanced stage as an increasing cavity (excavation).
Anatomie
Anatomically, a distinction is made between
open-angle and narrow-angle glaucoma. This classification is based on
the angle between the posterior surface of the cornea and anterior
surface of the iris. In the so-called iridocorneal angle is the
trabecular meshwork, through which the aqueous humour leaves the eye via
Schlemm’s canal.
Open-angle glaucoma is far more common and usually
happens over many years, is pain-free, and therefore often remains
unnoticed, while the rarer narrow-angle glaucoma can lead to a painful
glaucomatous attack, and if left untreated, there is the threat of
blindness within a short period of time.
Frequency
In
Switzerland, glaucoma is one of the most common causes of blindness.
Two in one hundred people aged 40 or over develop glaucoma. Evidently,
50,000 Swiss people suffer from glaucoma caused by increased intraocular
pressure, 10 percent of whom are threatened with blindness. Patients
with glaucoma with normal intraocular pressure (normal pressure
glaucoma, approx. 30 - 45% of all glaucoma cases) are not included in
this study. The Swiss Ophthalmological Society (SOG) points out that the
estimated number in this area is very high. It is assumed that in
Switzerland a total of about 100,000 people are affected by glaucoma.
Intraocular pressure
Aqueous
humour is produced In the ciliary body of the eye and delivered to the
posterior chamber of the eye. It passes through the pupil into the
anterior chamber of the eye and flows through the trabecular meshwork
via Schlemm’s canal. The ratio of aqueous humour production to aqueous
humour flow is determined by the intraocular pressure.
Normal eye
pressure is between 10 and 21 mm Hg, fluctuations of up to 5 mm Hg
during the day are normal. Intraocular pressure increases with age in
Europeans.
Causes
One cause of open-angle glaucoma is a chronic circulatory disorder of the optic nerve, which is frequently caused by increased intraocular pressure. The cause of increased intraocular pressure is impaired drainage of the aqueous humour from the anterior eye chamber. This results in a pressure increase in the eye, which in turn leads to increased pressure on the sensitive optic nerve fibres. The optic nerve head, the point at which all nerve fibres of the retina converge and leave the eye in the direction of the brain, is particularly sensitive. If the nerve fibres are no longer supplied with oxygen and other nutrients at this point, they will die if the underprovision is long enough.
With adequate blood circulation in the blood vessels of the
optic nerve head, higher intraocular pressure can be tolerated; with low
blood circulation, low intraocular pressure can lead to glaucomatous
progression (normal pressure glaucoma). Particularly unfavourable is the
combination of high intraocular pressure and low optic nerve head blood
flow. Since intraocular pressure as well as blood circulation can
fluctuate, both the absolute value of both parameters as well as the
duration of unfavourable pressure or blood circulation ratios are
significant.
All the conditions which, on the one hand,
contribute to increased intraocular pressure or, on the other hand, to
reduced optic nerve head blood flow, can hence cause glaucoma. Among the
former are notably disorders in the functioning of the trabecular
meshwork, (too) low blood pressure or over-treated hypertension, and
other diseases that restrict the blood circulation (arteriosclerosis,
cardiac arrhythmia and diabetes, etc.).
In particular, phases of a severe night-time drop in blood pressure can contribute to a worsening of the disease.
Neurodegeneration and oxidative stress also influence the incidence of the disease.
Individual combinations of these factors define the critical threshold, which determines glaucomatous progression.
The
damage to the optic nerve with glaucoma, visible in the characteristic
erosion of the optic nerve head, first affects the nerve fibres of the
middle peripheral retina, and progresses slowly towards the centre. When
approximately 70% of the nerve fibres are affected, arcuate
restrictions in the field of vision form in the middle field of vision,
which are often not perceived by the patient even at an advanced stage.
Intraocular
pressure can be recorded with different methods (Goldman applanation
tonometry or pneumotonometry). In this case, the force required for
defined flattening (mechanically or by a blast of air) of the cornea,
anaesthetised beforehand, is measured. The measurement result is
influenced by the patient’s corneal thickness. In the case of a thicker
cornea, higher values than actually present are measured; in the case of
a thinner cornea, lower values than actually present are measured.
Hence, an additional measurement of corneal thickness is required, and
the actual intraocular pressure has to be calculated from the raw data
of the measured pressure values via a corneal-specific correction
factor. Since intraocular pressure fluctuates during the day, several
measurements may be required spread over the day and possibly night (day
and night pressure profile).
Solely measuring the intraocular
pressure is not sufficient for making or excluding a diagnosis or
exclusion, or for assessing its course. The following additional
examinations are necessary:
Measurement of the blood circulation in the optic nerve
Measurement
and assessment of the fundus oculi in a three-dimensional image: The
extent of the damage to the optic nerve is determined by the size and
shape of erosion of the optic nerve head. The actual value of these
examinations is in the reproducible examination of progression which
allows early adjustment to treatment
Examination of the field of vision: we seek characteristic, arcuate (typical glaucoma) blind spots
Primary open-angle glaucoma
Open angle glaucoma is referred to as primary open-angle glaucoma, if its onset is not as a consequence of another eye disease.
Primary chronic open-angle glaucoma
Primary
chronic open-angle glaucoma is the most common form of glaucoma.
Typically, it occurs at the age of 40 and over, but can begin earlier.
Familial accumulation, i.e. genetic predisposition, is known. With this
form of glaucoma, a drainage obstruction occurs directly in the drainage
area of the iridocorneal angle as a result of degenerative changes.
Pressure in the eyeball increases slowly over years, and the patient
usually has no symptoms.
A particular form of primary chronic
open-angle glaucoma is normal pressure glaucoma. Here there is an onset
of progressive optic nerve damage, despite predominantly normal
intraocular pressure values. As a result of various factors, local optic
nerve head blood flow is so restricted that the optic nerve fibres are
also damaged.
Congenital (juvenile) glaucoma
Developmental
defects of the iridocorneal angle during the embryonic phase lead to a
drainage disorder of the intraocular fluid. This can occur in
combination with other malformations of the body. Increased intraocular
pressure can lead to an enlargement of the eyeball on one or both sides.
If congenital glaucoma is suspected, an examination, intraocular
pressure measurement and possibly, a timely operation must be carried
out at an early stage in order to prevent damage to the optic nerve.
Secondary open-angle glaucoma
The
onset of open-angle glaucoma as a consequence of other diseases is
known as secondary open-angle glaucoma. This is the case with injury to
or inflammation of the eye, intraocular tumours, neovascularisation of
the iridocorneal angle, for instance, due to diabetes or in the case of
predisposed persons, after taking certain drugs (e.g. cortisone),
so-called steroid responders.
Narrow-angle glaucoma
Narrow-angle
glaucoma results from a drainage disorder outflow disturbance of the
intraocular fluid as a consequence of a narrow angle between the iris
and the cornea, i.e. in front of the trabecular meshwork. The extent of
narrowing can fluctuate, for example, by the change in the pupil width
and hence the thickness of the iris. The drainage disorder results in
periodic or permanent increased intraocular pressure, which eventually
leads to optic nerve damage. People with higher long-sightedness
(shorter eye: narrower iridocorneal angle) and advanced cataracts
(thicker lens) are at risk. Certain drugs (anticholinergics – e.g. some
antidepressants or antiemetics) can lead to pupillary dilation and hence
to a rise in intraocular pressure and possibly to a glaucomatous
attack.
Glaucomatous attack
A
glaucomatous attack is based on a sudden reduction in intraocular fluid
drainage due to the displacement of the iridocorneal angle by the iris.
As a consequence, the intraocular pressure increases sharply, and the
affected eyeball often feels rock-hard to the touch. Typical symptoms
are sudden loss of vision, haloes (rainbow-coloured rings around
lights), severe headache, nausea, vomiting and possibly cardiac
arrhythmia. Usually only one eye is affected. The attack can abate after
a few hours without any treatment and return at intervals, but also
last for days undetected.
Treatment
If
glaucoma-typical damage to the optic nerve is detected, permanent
reduction in intraocular pressure must occur. Generally, damage to the
optic nerve is caused by a chronic excess of critical intraocular
pressure, insufficient supply of nutrients (impaired blood flow),
neuro-degeneration and / or increased oxidative stress. The critical
intraocular pressure varies individually and must be found individually
over the course of the disease through close-knit checks, and should
preferably not be exceeded long term by the appropriate treatment. In
addition, the optic nerve blood flow should be optimised, concomitant
diseases adjusted (internal co-treatment), neurodegenerative components
determined and oxidative stress reduced. The aim of the therapy is to
prevent the disease from progressing – damage that has already occurred
(field of vision defects) is irreversible.
The treatment begins
with eye drops (active substances: beta-blockers, cholineergics, alpha
sympathomimetics, carbonic anhydrase inhibitors and prostaglandins)
which reduce the intraocular pressure alone or in combination, with
different therapeutic approaches, and partly improve the optic nerve
blood flow.
In the case of secondary glaucoma, therapy for the underlying disease may be necessary in addition.
If
no satisfactory adjustments can be made in reducing intraocular
pressure or to blood flow with drugs, a surgical procedure suitable for
the patient (laser, surgical) must be quickly selected.
In
addition, the intraocular pressure can fall after surgery for cataracts
has been performed, since the plastic lens used is less thick than the
patient’s own lens, and hence indirectly provides space at the
iridocorneal angle.
Preventative screening
Regular
preventative screening at your ophthalmology centre is necessary. The
aim is to detect the disease at an early stage, i.e. before any
functional impairment. Timely treatment can prevent its progression and
ultimately the onset of blindness in most cases. From the age of 40,
preventative screening should be carried out once a year. Earlier
preventative screening is sensible with the following risk factors:
- Reduced blood circulation in the optic nerve - often with circulatory disorders (vasospasm) of the limbs (cold hands / feet), migraine, tinnituss
- Relatively or absolutely elevated eye pressure
- Advanced age
- High short-sightedness (open-angle glaucoma)
- High long-sightedness (narrow-angle glaucoma or glaucomatous attack)
- Low and fluctuating blood pressure (normal pressure glaucoma)
- High blood pressure
- Familial predisposition
- Diabetes
Further information
Further information can be found on our website and from the web links and references listed below.
Web links
http://www.glaukompatienten.ch
Patient information on glaucoma and guidelines by the Professional Association of Ophthalmologists (BVA) and the German Ophthalmological Society (DOG)
www.glaukom.de – Initiative group for the early detection of glaucoma
German Green Cross (GDK): Glaucoma and free glaucoma helpline
References
Books
Age-related macular degeneration (AMD)
PD Dr. med. Karl-Georg Schmidt
Definition of AMD
Age-related
macular degeneration (AMD) is a progressive, degenerative disease of
the eyes, whereby the location of sharpest vision, the macula ("yellow
spot") is damaged.
AMD is the main cause of the serious loss of vision (visual acuity) in the western population over the age of 65.
A healthy macula allows sharp vision as well as colour vision.
There
is to date no treatment that can cure this disease. Frequently,
however, progression of AMD can be prevented or at least delayed by
suitable treatment, which is dependent on the type of AMD and the stage.
Basically,
there are two distinct types of AMD; the milder, dry (non-neovascular,
i.e. without vascular regeneration) AMD and the more aggressive, wet
(neovascular, i.e. with vascular regeneration) AMD .
Commonly
preceding both is the loss of a certain cell layer, the retinal pigment
epithelium, which controls the nutrition of the cells of the macula.
Basics of AMD
The
macula of a healthy eye consists of photoreceptors, a layer located
over the retinal pigment epithelium (RPE) and the choroid (Figs 1 &
2). The choroid is vascular-rich and supplies the macula with oxygen and
nutrients. This occurs via the retinal pigment epithelium, which
transports oxygen and nutrients from the choroid into the macula and
removes toxic metabolic end products (cell waste).
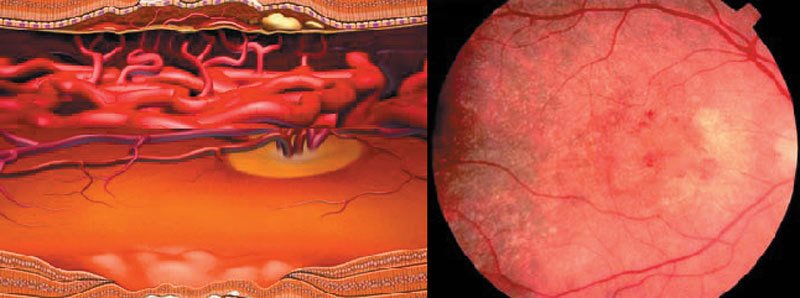
Left: Schematic representation of a cross-section through the normal outer retina with photoreceptors, retinal pigment epithelium (RPE) and choroid.
Right: Schematic representation of normal findings with healthy macula
Over the course of your life, the number of cells in the retinal pigment epithelium decreases.
If too many cells are destroyed in the retinal pigment epithelium (preliminary stage of AMD), the retinal pigment epithelium can no longer transport sufficient oxygen and nutrients into the macula, sufficient toxic metabolic end products are no longer transported away, the cells of the macula “starvation” and are "poisoned", and (dry) AMD starts. The earlier the corresponding therapy (see below) is started, i.e. the fewer sensory cells have perished at the start of treatment, the more likely AMD and hence deterioration of vision (with usually significant consequences with regard to quality of life) can be stemmed or delayed.
As already mentioned, since AMD cannot be healed, but can frequently be stemmed, early diagnosis and therapy is particularly important.
Cause of AMD
The
exact cause of AMD is not yet known. Probably several factors need to
converge for AMD to develop. From the age of 50, the probability of
developing AMD increases.
Decisive for the dying off of macula sensory cells is the decreasing supply of oxygen and nutrients and the accumulation of toxic metabolic end products. The cause of this is the destruction of the retinal pigment epithelium as a result of increased oxidative stress (increased formation of free radicals). If a correspondingly large number of cells of the retinal pigment epithelium have been destroyed, this cell layer can no longer adequately transport oxygen and nutrients into the macula, and no longer completely remove the waste products from macular metabolism. This results in the depositing of these waste products under the retinal pigment epithelium in the area of the macula (drusen), the nutrient situation in the macula increasingly deteriorates, the macula cells "starve", are "poisoned" and die off, and (dry) AMD starts.
Dry AMD
Dry
AMD starts with the loss of the retinal pigment epithelium, which
precedes the restriction of vision. Gradually, deposits under the
retina, the so-called drusen, increase in number and size, and blood
vessels supplying the macula perish (microcirculatory disorder). As a
consequence, the sensory cells of the macula die off. When cell loss and
deposits reach a critical limit, vision noticeably deteriorates. The
final stage is so-called geographic atrophy with a massive loss of
vision.
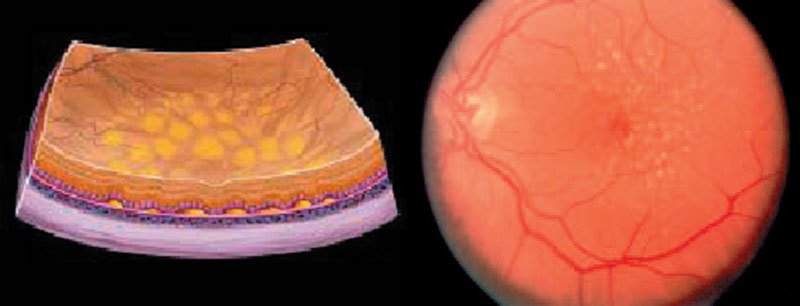
Left: Schematic representation of the retinal changes in an eye with dry (non-neovascular) AMD. Drusen (unremoved yellowish waste products from macular metabolism as a result of dying off (oxidative stress due to nutrient-deficiency-induced increased formation of free radicals) of the retinal pigment epithelium under the macula.
Right: Fundus image of an eye with dry (non-neovascular) AMD with drusen
Even if the disease initially only affects one eye, there is a risk that the other eye is also likely to become diseased within a few years.
Dry AMD can go on to become a wet form in 20 - 30% of affected patients. Accordingly, regular check-ups in agreement with your ophthalmologist are important (at least once or twice a year).
Wet AMD
The
transition from dry to wet AMD is characterised by the onset of
uncontrollably growing, abnormal choroidal neovascularisation (CNV –
images 5 & 6). The basic process, whereby new blood vessels form, is
called angiogenesis.
Angiogenesis is a regulated process, whereby new blood vessels are formed by the branching and extension of existing blood vessels. Angiogenesis plays an important role in many normal (physiological) processes such as wound healing, oxygen- deficiency-induced (hypoxic) damage and reproduction. With some diseases, for instance, cancer, arthritis, diabetes mellitus, and eye diseases such as wet (neovascular, i.e. with vascularisation) AMD, however, there is persistent and impaired (deregulated) angiogenesis.
Angiogenesis is characterised by a cascade of events. Local degradation of extracellular tissue is followed by blood vessel dilation and increased blood vessel permeability. This allows activated and proliferating endothelial cells to migrate (migration) and form tubes. It is assumed that these “sprouting” blood vessels are supported by a network of sophisticated periendothelial cells and extracellular matrix, before maturation and remodelling of the new blood vessels occurs (Carmeliet et al, 1996; Folkman & Shing, 1992).
The initial factor for angiogenesis in the eye is oxidative stress (nutrient-deficiency-induced increased formation of free radicals), which stimulates the upregulation and expression of vascular endothelial growth factor A (VEGF-A) and other angiogenic factors. This results in proliferation and migration of the endothelial cells, proteolysis and penetration of the basal membrane, followed by penetration of the blood vessels into the macula. Vascular endothelial growth factor A (VEGF-A) is an essential component of the angiogenic cascade and hence an important target for the treatment of certain stages of wet AMD (Das & McGuire, 2003; Witmer et al, 2003).
VEGF-A is a growth factor with different characteristics; it promotes cell division of endothelial cells, it is crucial for the survival of newly-formed blood vessels and it is a chemotactic factor for inflammatory cells. In addition, it is still known under a different name - vascular permeability factor (VPF), since VEGF-A regulates the permeability of blood vessels.
VEGF-A has an affinity for two VEGF-A receptors (VEGFR) located on the surface of endothelial cells: VEGFR-1 and VEGFR-2 (Keyt et al., 1996; Ferrara and Davis-Smyth, 1997). The binding of VEGF-A to these receptors leads to the growth of endothelial cells and to changes in the connections between endothelial cells (so-called "tight junctions", Aiello et al, 1995; Ozaki et al., 2000), thereby further reducing the impermeability of the blood vessels (Antonetti et al, 1999).
If these processes are not regulated, abnormal blood vessels grow through the retinal pigment epithelium from the choroid. These abnormal new blood vessels secrete fluid and / or blood, thereby detaching the retinal pigment epithelium. This can in turn manifest as a distorted view (metamorphopsia), a central blind spot (central scotoma) or inability to read with the diseased eye.
As a consequence of the leakage of blood and fluid from choroidal neovascularisation under the macula, a scar forms and thereby extensive and irreversible loss of function in the affected tissue (loss of vision).

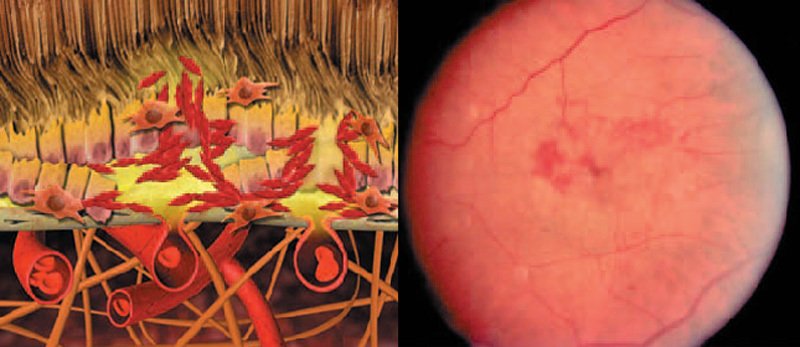
Left: Schematic representation of retinal changes and fundus image of an eye with wet (neovascular, exudative) AMD. Characteristic of wet AMD is uncontrolled growth of abnormal choroid blood vessels under and into the macula.
Right: Fundus image of an eye with wet (neovascular, exudative) AMD with blood and fluid discharge from abnormal choroid blood vessels
Left: Schematic representation of retinal changes in an eye with vascular leakage and disc-like scarring with wet AMD.
Right: Fundus image of retinal changes in an eye with vascular leakage and disc-shaped scarring with wet AMD
Development of AMD
The development of AMD depends on whether the AMD is dry or wet.
Wet AMD is the form that threatens our sight more severely. Development is more rapid and more aggressive. However, this form is also much rarer: approx. 5% of those aged over 60 are affected by wet AMD.
Dry AMD usually develops more slowly, sometimes over years. Dry AMD can go on to become wet AMD.
The progression of AMD is complex and can lead include transition from dry (non-neovascular) to wet (neovascular, exudative) AMD with the occurrence of choroidal neovascularisation (CNV) and the associated anatomical changes in the retina and retinal pigment epithelium.
The formation of a disc-shaped scar is the result of the leakage of blood and fluid from choroidal neovascularisation under and into the macula (Figures 7 & 8). This scar usually manifests as a white or yellowish-white, fibrous, generally round, raised lesion. Fresh bleeding at the edge of the scar indicate recurrent or persistent choroidal neovascularisation.
The progression of AMD
can be rapid. In clinical trials, 2.9% of untreated patients were unable
to read at the start, after 3 months the number increased to 13.4%, and
after 24 months to 46.3% (Macular Photocoagulation Study Group, 1991).
During the development of AMD, the visual impression becomes increasingly distorted and blurred in the central field of vision.
In around 40% of patients with wet AMD in one eye, this disease is likely to develop in the other eye over a period of 5 years (Macular Photocoagulation Study Group, 1993).
Reading, recognising faces and everyday tasks such as shopping or driving can no longer be handled with advanced AMD, and capacity for self-determination is reduced.
Vision survives in the outer area of the field of vision, as the disease does not lead to complete blindness.
Regular check-ups are important in order to detect AMD early on, ideally before the onset of any functional restriction, but also for timely detection of any deterioration or transition from dry to wet AMD.
Early signs of AMD
Initially,
the patient usually does not notice anything, as AMD develops in a
painless manner. Visual acuity of the affected eye is initially only
slightly limited or not at all, although there may already be a
significant and advanced loss of the retinal pigment epithelium (AMD
precursor). Even if vision is reduced, that is, if the disease is
already noticeable through functional loss in one eye, the healthy eye
often compensates for the loss in the diseased eye, so affected patients
often only notice AMD at a late stage.
A regular check of the back of the eye (fundus oculi) by a macula specialist is therefore particularly important from the age of 50. The medical skill consists of early detection of the slightest changes in the retinal pigment epithelium, exclusion of possible other causes, diseases and / or degeneration, and carrying out the best possible therapy in the affected patients at an early stage, preferably before the onset of functional impairment, i.e. before a decrease in visual capacity, so as to prevent progressive loss of vision. The sooner AMD is detected, the better the chances of stemming the progression of this disease by means of a suitable individual form and stage-dependent therapy with or without only slight visual impairment.
Onset of loss of vision is usually irreversible, since by this stage of the disease the sensory cells necessary for the visual process have already died off.
Signs of advanced (clinically manifest) AMD
Blurred, unsharp vision
Distorted images (so-called metamorphopsia)
Straight lines appear curved
Increasing problems reading and adjusting to darkness
Colours appear weaker
Restricted
visual perception (grey spot) or lack of visual perception (blind spot)
in the centre of the field of vision (so-called scotoma)
Risk factors for AMD
Age
The prevalence of AMD increases rapidly with age (Framingham-Study, Beaver Dam Eye Study, Netherlands Rotterdam Eye Study).
From the age of 60, approximately one in four people suffers from dry AMD. Around 18% of people between the ages of 70 and 74 have a form of AMD. 47% of people over the age of 85 have AMD. In approximately 90% of cases, dry AMD is present. In the case of people aged between 55-64, 0.1% has a wet AMD, and this figure increases to 7% from the age of 85 (Ferris et al, 1984; Klein et al, 1992; Vingerling et al, 1995).
Genetics
AMD is,
at least partially, an inherited condition, as studies with an
increased rate of disease in first-degree relatives of AMD patients
showed (Klein et al, 1992). If a first-degree relative suffers from AMD,
the other family members also have an increased risk of developing AMD.
Gender
Considering
all forms of AMD, men and women are affected more or less equally, but
the wet form of AMD is more common in women, especially over the age of
75 (Klein et al, 1997).
Smoking
Depending on the dose, cigarette smoke increases the risk of developing AMD.
Smokers
currently smoking around 20 or more cigarettes a day have a 23-fold
increased risk of developing AMD with vision loss compared to people who
have never smoked (AREDS Study Research Group, 2000).
Ethnic origin
AMD is more common in Caucasians than in Asians or African Americans (Klein et al, 2003).
Iris colour
There is evidence that people with a light-coloured iris are more prone to developing.
Nutrition with few antioxidants
The
prevalence rate of AMD is about twice as high in patients who only
consume small amounts of antioxidants and lutein compared to patients
who consume a high proportion of antioxidants and lutein (Oshinskie,
1996; AREDS Study Research Group, 2001 – active ingredients, see above).
High cholesterol values (Vingerling et al, 1995)
Hypertension (AREDS Study Research Group, 2000)
Cardiovascular disease (AREDS Study Research Group, 2000)
UV light (Nilsson et al, 2003; Rezai et al, 2008)
Quality of life with AMD
Although
AMD rarely causes complete blindness when it occurs in both eyes, the
ability of patients to deal with normal visual requirements such as
reading, handling money, reading the time, recognising faces and
driving, is severely impaired or no longer possible. As a result, the
quality of life of the affected patients is also significantly reduced.
Loss of vision that leads to a loss of independence (visual impairment
can reduce mobility and increase the risk of injuries, e.g. caused by a
fall) and lower self-esteem, limits patients very much and can cause
depression.
Estimations indicate that slight AMD causes a 17% reduction in quality of life. For moderately severe and severe forms of AMD, the quality of life is reduced to 40% or by more than 60% (Brown et al, 2005).
AMD is described by patients as just as restrictive as other chronic obstructive diseases such as arthritis, chronic obstructive pulmonary disease and AIDS (Acquired Immune Deficiency Syndrome) (Williams et al, 1998; Brody et al, 2001).
Investigations with AMD
With
suspected AMD, an examination of the macula is necessary. These
painfree examinations are carried out after dilation of the pupil (eye
droplets) with certain equipment (slit lamp, magnifying glasses,
ophthalmoscope). If certain changes (Figures 3 - 8) are displayed, e.g.,
pathological deposits of typically yellowish fat-containing degradation
products (drusen), pathological blood vessel sprouting or the leakage
of blood and / or fluid, i.e. indications of AMD, further investigations
may be necessary as follows:
Optical coherence tomography (OCT, to determine the retinal condition and retinal thickness)
Fundus diagnostic documentation (photography)
Vascular imaging (with fluorescein or indocyanine green angiography to highlight the blood vessels).
As a rule, first of all fundus diagnostic documentation is gathered with a special image (optical coherence tomography, OCT), then fluorescence angiography is carried out. Fluorescence angiography, a special dye examination, helps in distinguishing the AMD (form and stage), and abnormal vascular growth can be detected and assessed. If a specific form of wet AMD is suspected, vascular imaging with another dye, indocyanine green, may be helpful.
Optical coherence tomography, a painless, three-dimensional laser method for contactless, high-resolution layer examination, allows microstructural changes in the macula to be detected. By combining angiography and optical coherence tomography, the macula specialist can identify certain diseased structures in the living eye and hence precisely calculate and analyse the type and extent of the diseased deposits of metabolic end products. This allows the individual progression of AMD to be estimated and the therapeutic success to be checked.
Treatment of AMD
AMD is so far incurable, and there is no guaranteed protection against it.
The most important aim in the treatment of AMD is to stem the natural progression of the disease (early, ideally before the onset of visual loss, i.e. at the start of changes in the retinal pigment epithelium) and to preserve as much visual faculty as possible.
The intake of specific, high-quality non-heat-stabilised active ingredients with high bioavailability (DL-alpha-lipoic acid, zeaxanthin, lutein, levocarnitine, pantothenic acid, pyridine-3-carbamide, thiamine, riboflavin, 3-hydroxy-2-methylpyridine, vitamin B6, pteroyl-glutamic acid, biotin, cyanocobalamin) has proved effective here in non-smokers (Age-Related Eye Disease Study Research Group, 2007; Briganti et al, 2008; Feher et al, 2005; Kim et al, 2008; Johnson et al, 2008; Parisi et al, 2008; Zulkhairi et al, 2008)
In addition, the treatment of AMD depends on the existing form (dry or wet AMD) and the stage of disease.
Treatment of dry AMD
In the case of dry AMD, the microcirculation of the retina, inter alia, is impaired, (see above). This leads to an insufficient supply of oxygen and nutrients to the macula. In order to maintain the functional capacity of the macula and hence central vision, the lifelong supply of specific nutrients and active ingredients (see above) as well as a sufficient blood flow in the fine blood vessels under the macula are necessary. In the case of dry AMD, progressive loss of the retinal pigment epithelium and a marked worsening of macular blood flow occur as duration of the disease increases. Directly below the macula, deposits of metabolic degradation products (so-called drusen) occur due to loss of function in the retinal pigment epithelium, nutrient supply and hence the function of the macula is further restricted, central vision worsens increasingly. In the late stage of dry AMD, the sensory cells in the macula perish irretrievably as a result of the dying off of the retinal pigment epithelium (so-called geographic atrophy), and central vision deteriorates considerably.
With certain early forms of dry AMD, rheopheresis may be helpful in addition to the intake of the above-mentioned active ingredients.
Rheopheresis with AMD
Scientific studies have shown that improving the flow characteristics of blood can help patients with dry AMD over and beyond therapy with specific active ingredients (see above) (Brunner et al, 2000; Kirchhof 2004; Klingel et al, 2003; Klingel et al, 2006; Pulido et al, 2005).
This therapy is known as rheopheresis. The goal of rheopheresis is to stabilise and / or improve central vision.
Rheopheresis is treatment of the blood outside the body. The blood treatment corresponds to a filtration process which removes certain blood components, for example, some fats and proteins, which have an adverse effect on the blood flow characteristics. The patient then receives his own "purified" blood back. Foreign blood components are not used. There are a number of similar processes for blood treatment outside the body that have been routinely used in the context of renal and metabolic diseases for decades.
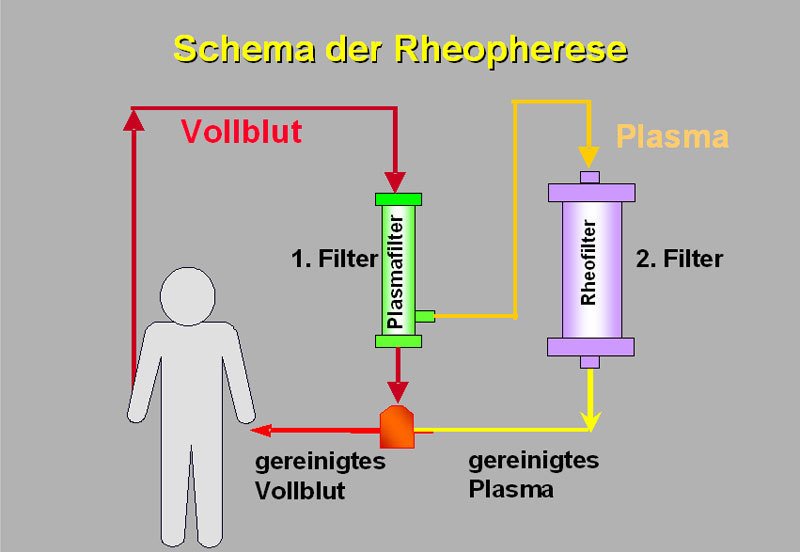
Schematic representation of rheopheresis
Rheopheresis treatment takes about 2-3 hours. During this time, the patient lies comfortably in bed. Two veins are punctured in the arms to create access to the bloodstream. The blood is piped from a vein through two filters into a tube system (see Figure 9). There is always only a limited amount of blood outside the body in the closed circuit. Tube system and filters are sterile disposable products. The purified blood is returned to the body via the second vein.
Treatment for wet AMD
In
the past, wet AMD was treated with laser photocoagulation, radiotherapy
or surgical procedures. The results of these methods were often
unsatisfactory. Newer pharmacological approaches, such as verteporfin
(Visudyne®) within the context of photodynamic therapy (PDT) or
pegaptanib sodium (Macugen®), an anti-VEGF-A165 RNA aptamer, resulted in
more constant results; frequently, stabilisation of vision was
achieved, which is defined as a loss of less than 15 letters on the
ETDRS (Early Treatment Diabetic Retinopathy Study) panel.
Newer VEGF antagonists (e.g. monoclonal antibody Fab fragments) specifically inhibit impaired (deregulated) angiogenesis, i.e. the basic mechanism for the growth of diseased blood vessels in the choroid. Correspondingly, the leakage of blood and liquid in the retinal pigment epithelium and the sensory cells (photoreceptors) leading to damage, scarring and then to irreversible visual loss, can be stopped or inhibited.
The mechanism of action is based on blocking the receptors of the vascular endothelial growth factor A (VEGF-A) on the surface of the endothelial cells, which form the choroidal blood vessels of the choroid membrane. In the case of wet AMD, the VEGF-A values are increased. Regulating this growth factor prevents, among other things, the cascade of processes which lead to the formation of choroidal neovascularisation and fluid leakage.
Current, randomised, controlled phase III studies (MARINA and ANCHOR) showed improved mean visual acuity during the first three months of treatment, which was able to be sustained over the entire treatment period. The cause seems to be the rapid and sustained reduction of retinal swelling and fluid leakage.
The natural progression of wet AMD to blindness (within the meaning of the law) can usually be prevented by this therapy.
The therapy begins with a phase of saturation (upload) of three intravitreal injections of the antiproliferative substance (VEGF inhibitor) into the affected eye at monthly intervals, followed by a maintenance phase, in which the visual acuity of the patients is checked monthly. Depending on the findings, further injections may be necessary.
An advantage of the injection into the
eye is that the active ingredient reaches the macula directly, i.e. the
place where it is intended to be effective. If the patient takes the
active ingredient in the form of tablets, a significantly higher dose
would have to be taken, since the macula only absorbs small amounts of
the substance from blood. In addition, the entire body would be exposed
to the higher dose of active ingredient, which could lead to side
effects.
The injection can be performed on an outpatient basis, but
must be performed under controlled, sterile conditions (in the operating
theatre), and the patient should use antibiotic eye drops 3 days before
and after the injection. An appropriate ophthalmologic check tailored
to the result should be carried out for a period of one week after the
injection, even in the absence of symptoms.
Vision may be blurred following the injection into the eye. After the procedure, the patient should not drive vehicles or operate machines as long as this symptom of blurred vision persists.
With signs of inflammation (reddening of the eyes, pain, sensitivity to light, or change in vision), the patient should immediately contact the treating macula specialist.
Preventative approach to AMD
There is no perfect protection against AMD.
However, the following behaviour patterns may prevent AMD:
Sunglasses with 100% UV protection and wide side pieces so that no UV light can penetrate your eyes from the side
Regular inspection of your ocular fundus
Self-testing of your eyesight regularly with the so-called Amsler grid
The corresponding active ingredients, Including antioxidants (not for smokers)
Cessation of smoking
Balanced diet (fruit, vegetables, fish)
Check of your blood pressure by your family doctor, with possibly a blood pressure adjustment
Check of your blood lipid profile by your family doctor, with possibly a reduction in your blood lipid levels
Any changes in your vision should be detected as early as possible and treated where possible.
As
already mentioned, AMD cannot be cured, but often stemmed or delayed.
Therefore early diagnosis and therapy is particularly important. If a
pre-stage of AMD or AMD is already manifest, a "healthy" diet alone is
insufficient and therapy is usually required.
If
you already have a clinically manifest AMD, it is important that the
transition from dry to wet AMD is detected at an early stage in order to
supplement or switch your treatment in good time.
Check-ups with AMD
You
should arrange your check-ups with your treating macula specialist. As a
rule, check-ups are required at least once or twice a year.
If you already have a wet form of AMD, you should usually go for a check-up every three to four months.
In the case of changes in your vision (Amsler test - see above), a prompt check-up should be carried out.
Selected references
- Adamis AP, Shima DT, Tolentino MJ et al (1996). Inhibition of vascular endothelial growth factor prevents retinal ischemia-associated iris neovascularisation in a nonhuman primate. Arch Ophthalmol; 114(1):66-71.
- Age-Related Eye Disease Study Research Group (2000). Risk-factors associated with age-related macular degeneration. A case-controlled study in the Age-Related Disease Study: AREDS report no. 3. Ophthalmology; 107:2224-32.
- Age-Related Eye Disease Study Research Group (2001). A randomised, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch Ophthalmol; 119(110):1417-36.
- Age-Related Eye Disease Study Research Group, SanGiovanni JP, Chew EY, Clemons TE, Ferris FL 3rd, Gensler G, Lindblad AS, Milton RC, Seddon JM, Sperduto RD (2007). The relationship of dietary carotenoid and vitamin A, E, and C intake with age-related macular degeneration in a case-control study: AREDS Report No. 22. Arch Ophthalmol; 125(9):1225-32.
- Aiello LP, Avery RL, Arrigg PG, et al (1994). Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med; 331(22):1480-7.
- Aiello LP, Northrup JN, Keyt BA, et al (1995). Hypoxic regulation of vascular endothelial growth factor in retinal cells. Arch Ophthalmol; 113(12):1538-44.
- Aiello LP, Brucker AJ, Chang S, et al (2004). Evolving guidelines for intravitreous injections. Retina; 24:S3-S19.
- Antiplatelet Trialists‘ Collaboration (1994). Collaborative overview of randomised trials of antiplatelet therapy–III: Reduction in venous thrombosis and pulmonary embolism by antiplatelet prophylaxis among surgical and medical patients. BMJ; 308(6923):235-46.
- Antonetti DA, Barber AJ, Holinger LA et al (1999). Vascular endothelial growth factor induces rapid phosphorylation of tight junction proteins occluding and zonula occluden 1. A potential mechanism for vascular permeability in diabetic retinopathy and tumors. J Biol Chem; 274(33):23463-7.
- Bird AC, Bressler NM, Bressler SB, et al (1995). An international classifi cation and grading system for agerelated maculopathy and age-related macular degeneration. The International ARM Epidemiological Study Group. Surv Ophthalmol; 39:367-74.
- Bonastre J, Le Pen C, Anderson P, et al (2002). The epidemiology. Economics and quality of life burden of age-related macular degeneration in France, Germany, Italy and the United Kingdom. Eur J Health Econ; 3(2):94-102.
- Bressler NM (2002). Early detection and treatment of neovascular age-related macular degeneration. J Am Board Fam Pract; 15(2):142-52.
- Briganti S, Wlaschek M, Hinrichs C, Bellei B, Flori E, Treiber N, Iben S, Picardo M, Scharffetter-Kochanek K (2008). Small molecular antioxidants effectively protect from PUVA-induced oxidative stress responses underlying fibroblast senescence and photoaging. Free Radic Biol Med; May 17.
- Brody BL, Gamst AC, Williams RA, et al (2001). Depression, visual acuity, comorbidity, and disability associated with age-related macular degeneration. Ophthalmology; 108(10):1893-1900.
- Brown DM, Kaiser PK, Michels M, et al (2006). Ranibizumab versus verteporfi n for neovascular age-related macular degeneration. N Engl J Med; 355:1432-44.
- Brown MM, Brown GC, Stein JD, et al (2005) Age-related macular degeneration: Economic burden and value-based medicine analysis. Can J Ophthalmol; 40(3):277-87.
- Brunner R, Widder RA, Walter P, Lüke C, Godehardt E, Bartz-Schmidt KU, Heimann K, Borberg H (2000). Influence of membrane differential filtration on the natural course of age-related macular degeneration: a randomized trial. Retina; 20(5):483-91.
- Carmeliet P, Ferreira V, Breier G, et al (1996). Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature; 380(6573):435-9.
- Ciulla TA, Danis RP, Harris A (1998). Age-related macular degeneration: A review of experimental treatments. Surv Ophthalmol; 43(2):134-46.
- Chen Y, Wiesmann C, Fuh G, et al (1999). Selection and analysis of an optimized anti-VEGF antibody: Crystal structure of an affi nity-matured Fab in complex with antigen. J Mol Biol; 293(4):865-81.
- Chidlow G, Schmidt KG, Wood JPM, Osborne NN (2002). Lipoic acid protects the retina against ischaemia/reperfusion. Neuropharmacology; 43:1015-25.
- Das A and McGuire PG (2003). Retinal and choroidal angiogenesis: Pathophysiology and strategies for inhibition. Prog Retin Eye Res; 22(6):721-48.
- De Jong PT (2006). Age-related macular degeneration. New Engl J Med; 5;355(14):1474-85.
- Ferrara N and Davis-Smyth T (1997). The biology of vascular endothelial growth factor. Endocr Rev; 18(1):4-25.
- Feher J, Kovacs B, Kovacs I, Schveoller M, Papale A, Balacco Gabrieli C ( 2005). Improvement of visual functions and fundus alterations in early age-related macular degeneration treated with a combination of acetyl-L-carnitine, n-3 fatty acids, and coenzyme Q10. Ophthalmologica; 219(3):154-66.
- Ferrara N, Damico L, Shams N, et al (2006). Development of ranibizumab, an anti-vascular endothelial growth factor antigen binding fragment, as therapy for neovascular age-related macular degeneration. Retina; 26(8):859-70.
- Ferrara N, Gerber HP, and LeCouter J (2006). The biology of VEGF and its receptors. Nat Med; 9(6):699-76.
- Ferris FL 3rd, Fine SL, Hyman L (1984). Age-related macular degeneration and blindness due to neovascular maculopathy. Arch Ophthalmol; 102(11):1640-2.
- Ferris FL, Kassoff A, Bresnick GH, et al (1982). New visual acuity charts for clinical research. Am J Ophthalmol; 94:91-6.
- Fine SL, Berger JW, Maguire MG, Ho AC (2000). Age-related macular degeneration. N Engl J Med; 342(7):483-92.
- Finger PT, Berson A, Sherr A, et al (1996). Radiation therapy for subretinal therapy for subretinal neovascularization. Ophthalmology; 103:878-89.
- Folkman J and Shing Y (1992). Angiogenesis. J Biol Chem; 267(16):10931-4.
- Friedman DS, Congdon NG, Kempen JH, Tielsch JM (2002). Vision Problems in the US: Prevalence of Adult Vision Impairment and Age-Related Eye Disease in America. 4th ed. Schaumburg, IL: Prevent Blindness America.
- Gaudreault J, Reich ME, Arata A et al (2003). Ocular Pharmacokinetics and Antipermeability Effect of rhuFab V2 in Animals. Invest Ophthalmol Vis Sci. 44:ARVO E-Abstract 3942.
- Gaudreault J, Fei D, Rusit J et al (2005). Preclinical pharmacokinetics of Ranibizumab (rhuFabV2) after a single intravitreal administration. Invest Ophthalmol Vis Sci; 46(2):726-33.
- Gehrs KM, Anderson DH, Johnson LV et al (2006). Age-related macular degeneration – emerging pathogenetic and therapeutic concepts. Annals of Med; 38(7):450-71.
- Goehring ( 2006). Incidence of myocardial infarction and cerebrovascular accidents in patients with wet age-related macular degeneration in a managed care population. Presented at 22nd International Conference on Pharmacoepidemiology & Therapeutic Risk Management, Lisbon, Portugal, 24-27 August.
- Gragoudas ES, Adamis AP, Cunningham ET Jr. et al (2004). Pegaptanib for neovascular age-related macular degeneration. N Engl J Med; 351(27):2805-16.
- Green WR (1999). Histopathology of age-related macular degeneration. Mol Vis; 5:27.
- Hammond CJ, Webster AR, Snieder H et al (2002). Genetic Influence on Early Age-related Maculopathy. Ophthalmology; 109:730-6.
- Hee MR, Puliafi to CA, Wong C, et al (1995). Quantitative assessment of macular edema with optical coherence tomography. Arch Ophthalmol; 113(8):1019-29.
- Heier JS, Antoszyk AN, Pavan PR, et al (2006). Ranibizumab for treatment of neovascular age-related macular degeneration: A phase I/II multicenter, controlled, multidose study. Ophthalmology; 113(4):633-42.
- Hoeller U, Fuisting B, Schwartz R, et al (2005). Results of radiotherapy of subfoveal neovascularization with 16 and 20 Gy. Eye; 19(11):1151-6.
- Houck KA, Leung DW, Rowland AM et al (1992). Dual regulation of vascular endothelial growth factor bioavailability by genetic and proteolytic mechanisms. J Biol Chem; 267(36):26031-7.
- Johnson EJ, Chung HY, Caldarella SM, Snodderly DM (2008). The influence of supplemental lutein and docosahexaenoic acid on serum, lipoproteins, and macular pigmentation. Am J Clin Nutr; 87(5):1521-9.
- Keyt BA, Nguyen HV, Berleau LT et al (1996). Identifi cation of vascular endothelial growth factor determinants for binding KDR and FLT-1 receptors. Generation of receptor-selective VEGF variants by site-directed mutagenesis. J Biol Chem; 271(10):5638-46.
- Kim JH, Kim JH, Yu YS, Jun HO, Kwon HJ, Park KH, Kim KW (2008). Inhibition of choroidal neovascularization by homoisoflavanone, a new angiogenesis inhibitor. Mol Vis; 14:556-61.
- Klein R, Klein BE, Moss SE. Diabetes (1992) Hyperglycemia, and age-related maculopathy. The Beaver Dam Eye Study. Ophthalmology; 99(10):1527-34.
- Klein R, Klein BEK, Jensen SC, Meuer SM (1997). The five-year incidence and progression of age-related maculopathy. The Beaver Dam Eye Study. Ophthalmology; 104:7-21.
- Klein R, Klein BE, Marino EK, et al (2003). Early age-related maculopathy in the Cardiovascular Health Study. Ophthalmology; 110:25-33.
- Klingel R, Fassbender C, Fassbender T, Göhlen B (2003). Clinical studies to implement Rheopheresis for age-related macular degeneration guided by evidence-based-medicine. Transfusion and Apheresis Science; 29: 71–84.
- Klingel R, Wong D, Fassbender CC, Heibges A, Erdtracht B (2006). Rheopheresis for Dry Age-Related Macular Degeneration (AMD)-RheoNet-Registry Update 2005/2006. ARVO Abstract: 2179.
- Kirchhof B (2004). Option für die Frühform der trockenen AMD – Rheopherese bei Altersabhängiger Makuladegeneration. Ophthalmologische Nachrichten; 6,1-5.
- Krzystolik MG, Afshari MA, Adamis AP, et al (2002). Prevention of experimental choroidal neovascularisation with intravitreal anti-vascular endothelial growth factor antibody fragment. Arch Ophthalmol; 120(3):338-46.
- Kvanta A, Algvere PV, Berglin L, Seregard S (1996). Subfoveal fibrovascular membranes in age-related macular degeneration express vascular endothelial growth factor. Invest Ophthalmol Vis Sci; 37(9):1929-34.
- La Cour M, Kiilgaard JF, Nissen MH (2002). Age-related macular degeneration: Epidemiology and optimal treatment. Drugs Aging; 19(2):101-33.
- Lafaut BA, Bartz-Schmidt KU, Vanden Broecke C et al (2000). Br J Ophthalmol; 84(3): 239-43.
- Leidy NK, Revicki DA, Geneste B (1999) Recommendations for evaluating the validity of quality of life claims for labelling and promotion. Value Health; 2(2):113-27.
- Lopez PF, Sippy BD, Lambert HM, et al (1996). Transdifferentiated retinal pigment epithelial cells are immunoreactive for vascular endothelial growth factor in surgically excised age-related macular degeneration choroidal neovascular membranes. Invest Ophthalmol Vis Sci; 37(5):855-68.
- Lowe J (2003). RhuFab V2 inhibits VEGF-isoforms-stimulated HUVEC proliferation. Ophthalmol Vis Sci; 44:E-Abstract 1828.
- Macular Photocoagulation Study Group (1991). Laser photocoagulation of subfoveal neovascular lesions in age-related macular degeneration. Results of a randomised clinical trial. Arch Ophthalmol; 109(9):1220-31.
- Macular Photocoagulation Study Group (1993). Five-year follow-up of fellow eyes of patients with agerelated macular degeneration and unilateral extrafoveal choroidal neovascularisation. Arch Ophthalmol; 111(9):1189-99.
- Macular Photocoagulation Study Group (1994). Laser photocoagulation for subfoveal choroidal neovascularisation. Five-year results from randomised clinical trials. Arch Ophthalmol; 112(4):500-9.
- Mangione CM, Lee PP, Gutierrez PR, et al (2001). National Eye Institute Visual Function Questionnaire Field Test Investigators. Development of the 25-item National Eye Institute Visual Function Questionnaire. Arch Ophthalmol; 119(7):1050-8.
- Mattice E, Wolfe CP (1986). Using the Amsler grid. J Ophthalmic Nurs Technol; 5(1):34
- McGraw P, Winn B, Whitaker D (1995). Reliability of the Snellen chart. BMJ; 310(6993):1481-2.
- Mordenti J, Cuthbertson RA, Ferrara N et al (1999). Comparisons of the intraocular tissue distribution, pharmacokinetics, and safety of 125I-labeled full-length and Fab antibodies in rhesus monkeys following intravitreal administration. Toxicol Pathol; 27(5):536-44.
- National Eye Institute, US NIH 2006, Age-related macular degeneration. Retrieved December 5, 2006, from http://www.nei.nih.gov/health/maculardegen/armd_fa...
- Nilsson SE, Sundelin SP, Wihlmark U, Brunk UT ( 2003). Aging of cultured retinal pigment epithelial cells: oxidative reactions, lipofuscin formation and blue light damage. Doc Ophthalmol; 106(1):3-6.
- Osborne NN, Chidlow G, Wood JPM, Schmidt KG, Casson R, Melena J (2001). Expectations in the treatment of retinal diseases: neuroprotection. Current Eye Res; 22:321-32.
- Osborne NN, Wood JPM, Chidlow G, Casson R, DeSantis L, Schmidt KG (2004). Effectiveness of levobetaxolo